Steam-liquid equilibrium apparatus (VLE)
Knowledge of the vapour-liquid equilibrium data (VLE) of a system is the basis for all distillative separation processes of liquid mixtures. Based on the equilibrium data, in combination with the mass balance of the system during discontinuous distillation, it is possible to predict the concentration curve as a function of the over-distilled quantity and thus the separation of the mixture into feed, distillate (possibly into several fractions) and residue. For continuous distillation or rectification, the theoretical number of stages, the corresponding concentrations and the reflux ratio can be determined.
In the case of ideal mixtures, the corresponding equilibrium concentration in the vapour for a given liquid concentration can be calculated simply by knowing the vapour pressures of the pure components. For real mixtures, however, activity coefficients are required for correction, the determination of which can be determined experimentally with the following apparatus. The equilibrium apparatus produces samples of the liquid and gas phases from the boiling mixture, which are then analysed, or the vapour-liquid characteristic is quantitatively determined by a combination of precision dosing equipment and measurement of the vapour pressure with high-precision pressure sensors.
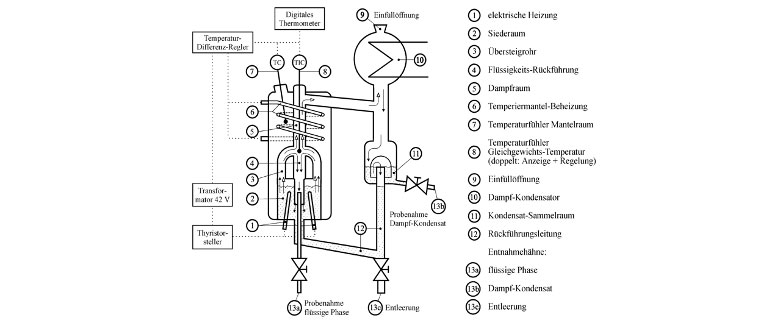
The liquid is dosed into the equilibrium apparatus via the filler neck (9) and fills the chambers (2), (11) and (12). The liquid mixture is brought to boiling point in the boiling chamber (2) by the two quartz immersion heaters (1). The vapour and liquid are conveyed together through the transfer tubes (3) into the vapour chamber (5). This path travelled jointly by the vapour and liquid during continuous mixing contributes significantly to achieving a state of equilibrium. This is not possible directly during the evaporation process in the boiling chamber, especially since no uniform concentrations can be expected there as a result of the condensate returned through the pipe (12) with a composition differing from the contents of the boiling chamber. The entrained liquid separates on entering the vapor space (5) and is returned through the pipe (4) below the liquid surface in the lower area of the boiling chamber. This “hydraulic closure” (4) prevents the vapor from directly entering the vapor space (5) by bypassing the transfer tubes (3). The ascending vapor has only a low velocity in the vapor space (5) due to its large cross-section which prevents the entrainment of liquid droplets. It is completely condensed in the condenser (10) with separation of inerts and returns to the boiling chamber through the return pipe (12). Stationary conditions with constant concentrations can be achieved by operating the circuit for a sufficiently long time.
The equilibrium occurs approximately at the separation of vapour and liquid after mixing in the transfer pipes at the entry into the vapour space. Therefore, the equilibrium data (boiling temperature, concentration of the liquid, concentration of the vapour) should be measured at this point, if possible:
- The temperature sensor (8 TIC) of the digital thermometer (platinum resistance thermometer) is located at the point of separation of steam and liquid at the inlet of the transfer tubes into the steam space.
- The samples of the liquid phase are taken with the aid of a capillary tube which reaches close to the point of separation of steam and liquid (sample tap 13 a).
- The condensate drain from the cooler (10) is used to sample the steam. The vapour and condensate concentration are the same if partial condensation of vapour in the vapour space is prevented. A condensate quantity of approx. 2 ml is present in the condensate collecting chamber (11) which can be drawn off and analyzed by the sampling valve (13b) after the stationary state has been reached.
Partial condensation of the steam in the steam chamber is prevented by the electrical heating coil (6). This protective heating is controlled by the differential temperature regulator, which compares the boiling temperature (8 TIC) measured at the point where steam and liquid separate, with the temperature in the jacket (7 TC) and maintains it at a previously adjusted difference.
The concentrations in the samples of the condensed vapour and liquid phase are determined by measuring the density or refractive index.
Through further highly accurate dosages, the complete concentration range of binary systems or sections of multi-component systems and thus the VLE data can be determined.


